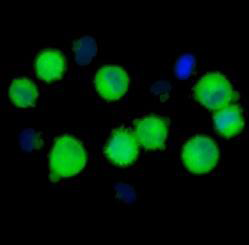
Nanotoxicology
“Impact of Nanomaterials on Human Health: Lessons from in vitro Models” is the currently completed project supported by a grant through the Norwegian Financial Mechanism and is devoted to examination of nanoparticle (NP) cytotoxicity , genotoxicity and oxidative stress in human cells in vitro. NP defined as particles having at least one dimension below 100 nm have been applied in the last decade in industry and medicine. Recently, there is an increased concern about the biohazard aspect of the presence of NP in consumer goods and in environment. Silver NP (Ag NP) cause in mammalian cells oxidative stress resulting from generation of ROS (reactive oxygen species), thus bearing some resemblance to indirect action of low LET ionizing radiation. The treatment with Ag NP brings about genotoxic and mutagenic effects, disturbed mitochondrial respiration, slowed proliferation and cell death.
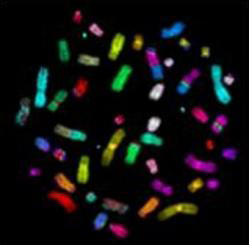
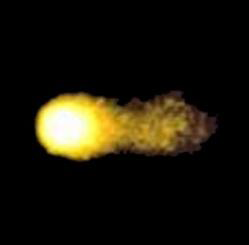
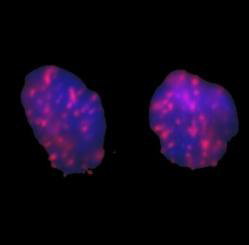
We studied the effects of DNA damage induced by treatment with uncoated silver NP (Ag NP) 20 nm or 200 nm Ag particles and titanium dioxide nanoparticles (TiO2 NP) 20 nm in 3 human cell lines: hepatocellular liver carcinoma HepG2, colorectal adenocarcinoma HT29 and lung carcinoma A549. The transmission (TEM) or scanning (SEM) electron microscopic images of these particles are shown in Figs 1-4. The endpoints examined were DNA breakage estimated by the comet assay and oxidative base damage recognized by formamido-pyrimidine glycosylase (FPG) and estimated with the FPG + comet assay, frequencies of histone γH2AX foci and micronuclei, apoptosis and clonogenic survival. An example of the comet assay determination of DNA damage is given in Fig. 5. Each cell line had a different pattern of DNA breakage and base damage versus NP concentration and time of treatment. There were no increases in the frequencies of histone γH2AX foci and micronuclei as compared to those in the untreated cells.
The reported experiments provided no data that would directly correlate DNA damage induced by Ag NP or TiO2 NP to early apoptosis or loss of clonogenic ability. This does not mean that DNA lesions induced by NP are harmless, especially at the organismal level. When incorrectly repaired, they may lead to mutations, in consequence bringing about various adverse effects on human health, including cancer predisposition, neurodegeneration, and immunodeficiency.
Another study concerned the effect of NP treatment on the repair of X-ray-inflicted DNA damage. Using alkaline comet assay, we examined the effect of combined treatment with Ag NP 20 nm or 200 nm Ag particles and X-rays (2 Gy) in HepG2 cells. In addition, combined treatment with X-rays and TiO2 NP 21 nm was also studied. No effect was observed of NP pre-treatment on X-ray induced initial DNA damage levels for all three particle types. In contrast, Ag particle treatment preceding exposure to X-rays caused a marked decrease in the rate of single strand break rejoining. The effect was particularly strong for Ag NP 20 nm. TiO2 NP pre-treatment had no effect on DNA repair (Fig. 6).
Both studies indicate that AgNP are by no means as safe as could be expected on the basis of early studies and their use should be carefully controlled.

Fig. 1. TEM image of TiO2 nanoparticles
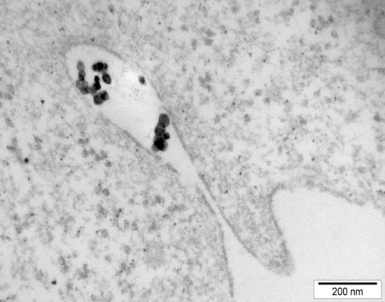
Fig. 2. Endocytosis of TiO2 nanoparticles in A549 cells
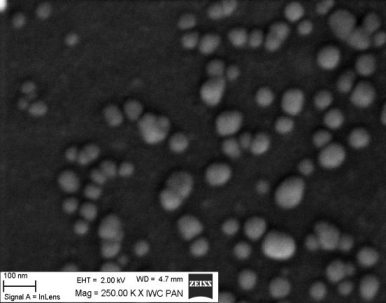
Fig. 3. SEM image of Ag nanoparticles
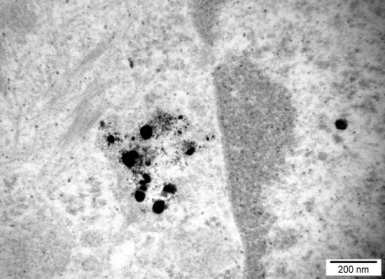
Fig. 4. Cellular distribution of Ag nanoparticles in the nucleus and cytoplasm of A549 cells
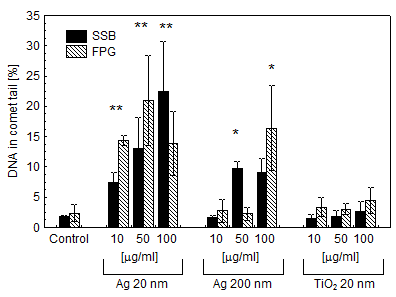
Fig. 5. DNA damage (single stand breaks, SSB, and base damage recognised by formamido-pyrimidine glycosylase, FPG) in HepG2 human hepatoma cells treated with Ag NP, Ag 200 nm particles and TiO2 NP for 2 h at concentrations indicated. Asterisks mark statistically significant difference from control (Student’s t test). Determination with the comet assay (three independent repeats) standard deviation indicated
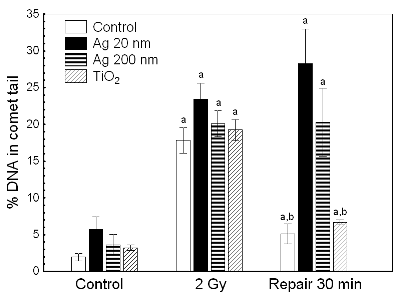
Fig. 6. The effect of nanoparticle pretreatment on repair of X-ray-induced DNA damage. Letter (a) denotes statistically significant difference vs control, letter (b) denotes statistically significant difference vs 2 Gy